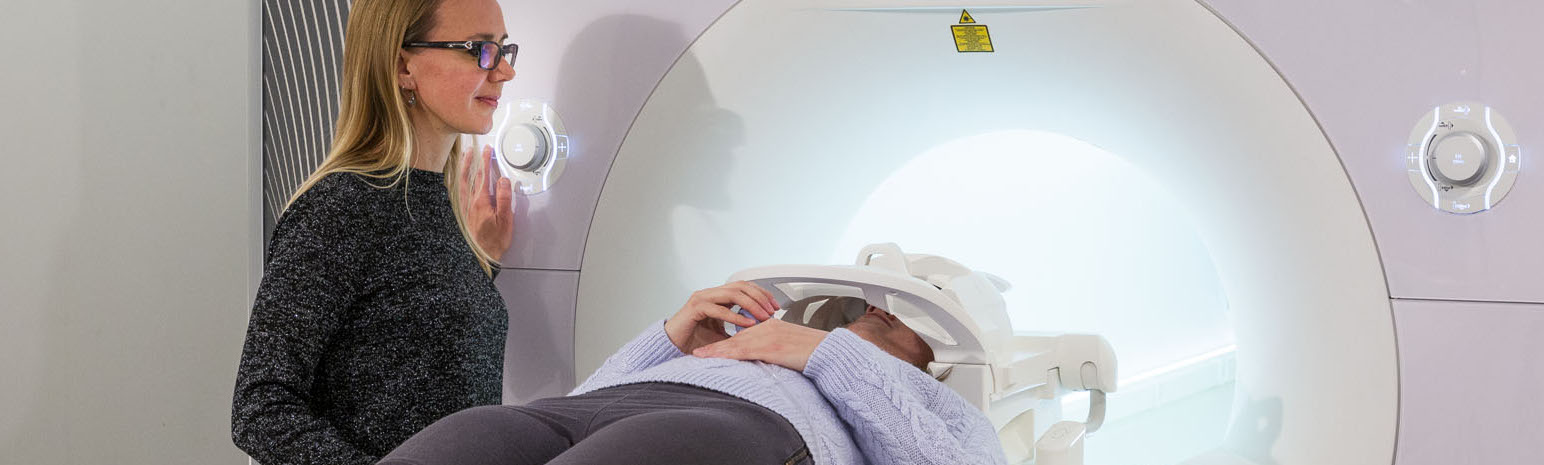
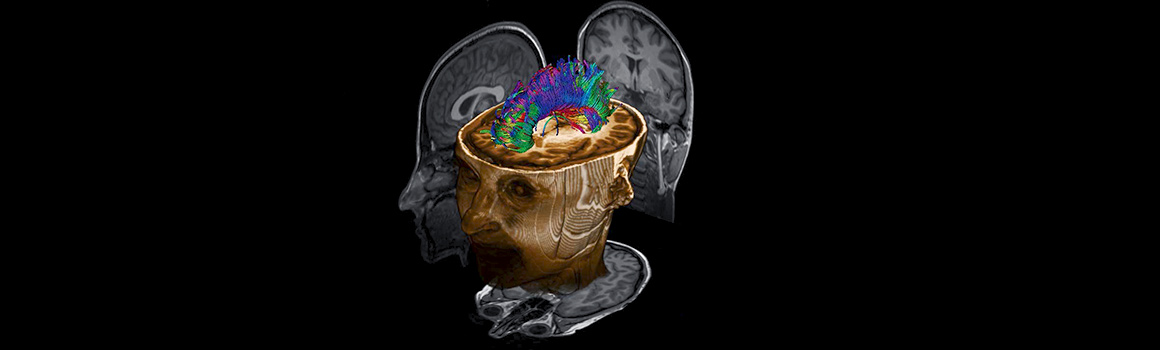
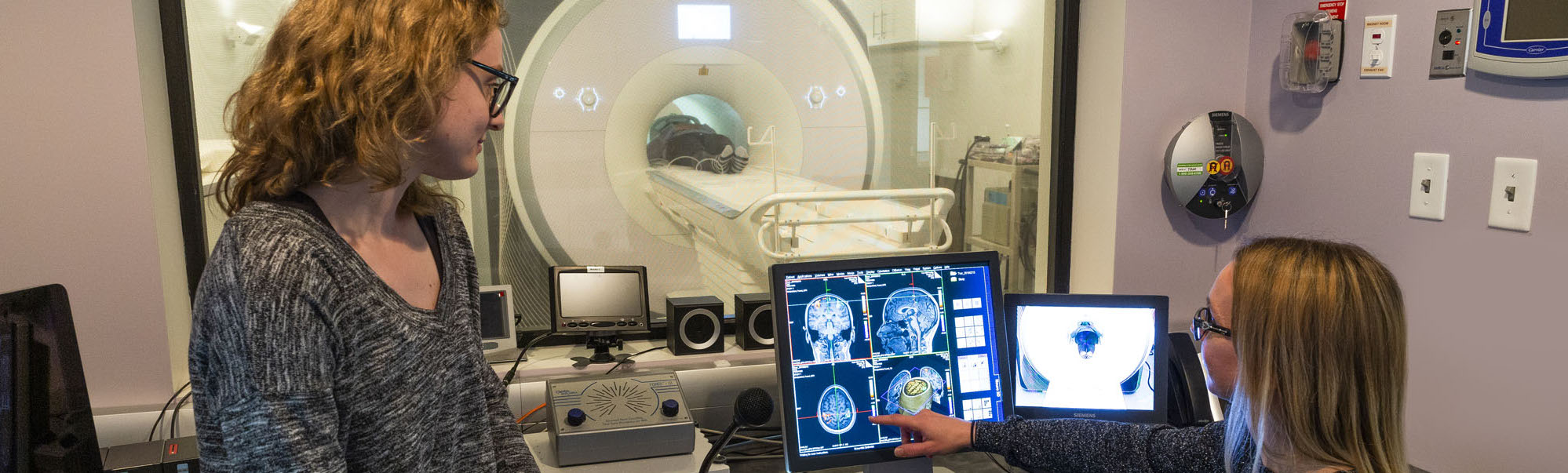
Our facility offers access to high field, full service MRI capacity at York University. We have a 3T Siemens Tim Trio with extensive MR compatible research tools and a full-time medical MRI Technologist. We support R & D phase medical device development and technology. We also support academic research with a specialty in neuroimaging. We allow extensive windows of scanner time enabling faster and more cost-effective turn-around for research, product development and testing.